In vivo research design
ARRIVE Pointers 2.0 have been adopted for research planning, conduct and writing. This was a single-arm pilot research to evaluate feasibility and security of tissue-engineered grafts to restore esophageal tissue defects. No management arm was used as there was adequate literature to recommend higher outcomes in cellularized grafts. Major final result was survival with secondary outcomes of 30-day mortality, intervention morbidity (anastomotic leak, an infection, endoscopic intervention, stricture and stent migration), perform (animal development, enteral autonomy and graft contractility) and tissue integration (histological, biomechanical and transcriptional similarity to native tissue). The pattern dimension was n = 8. The deliberate endpoint was 6 months, with predetermined humane endpoints, as additional detailed beneath. All animals have been included in evaluation, no matter endpoint. Major final result and primary secondary outcomes couldn’t be blinded. Histopathological evaluation (Figs. 4e,g,i,l and 5c,e, and Prolonged Information Fig. 4c,d), isometric contractility testing (Fig. 5a,b) and biomechanical evaluation (Fig. 3c) have been carried out blinded to postoperative survival and, the place relevant, stage of esophageal part (that’s, native or graft).
Animal husbandry
All in vivo work was undertaken at The Griffin Institute, Northwick Park Institute for Medical Analysis (institution license XA57A4134, PPL PP3815734). Feminine Göttingen minipigs (4–5 kg, 8 weeks previous, Ellëgard) have been transported to the power, acclimatized for 3 weeks earlier than experimental begin and examined for well being standing earlier than research inclusion. Distinctive identifiers have been marked on the outer side of 1 ear. Animals have been initially group-housed, singly housed after surgical procedure for as much as 7 days after which group-housed in concrete pens with rubber mats, heaters and environmental enrichment with a 12-h gentle–darkish cycle, ambient temperature (15–24 °C) and goal humidity 40–70%. Animals obtained water ad libitum all through the research, with particular minipig (SMP) pellets (220–280 g per day) and fruit treats (for instance, banana and apple) as enrichment. Physique weight was recorded on arrival, immediately preoperatively and weekly thereafter.
Cell isolation, growth and differentiation
Autologous minipig (n = 8, 3–4 months of age, 4.75–8.4 kg) MABs and FBs have been remoted from rectus abdominis muscle and fascial biopsies, respectively, as for human MABs, with minor modifications48. The biopsy (5 × 5 mm) was rinsed in PBS earlier than separation of fascia and muscle and dissection into 2 × 2-mm2 items. The items have been plated on 100-mm tissue-culture-treated dishes (Corning) coated with 1% Matrigel growth-factor-reduced basement membrane matrix (Corning) in 3 ml of proliferation medium (PM; Megacell DMEM (Sigma-Aldrich), 5% FBS (Gibco), penicillin–streptomycin resolution (P/S; Gibco), L-glutamine (Gibco), 2-mercaptoethanol (Sigma-Aldrich), MEM nonessential amino acid resolution (Sigma-Aldrich), 10 ng ml−1 bFGF (R&D Methods) and incubated at 5% CO2, 5% O2, 37 °C. After attachment (2–16 h), 7 ml of PM was gently added. After 3–5 days, first outgrowths have been indifferent by remedy with TrypLE Categorical (Gibco), transferred to 0.5% Matrigel-coated flasks and passaged each 2–3 days relying on confluence. Tissue was replated as much as seven occasions or till no additional outgrowths fashioned. For differentiation assay, cells have been plated on 0.5% Matrigel-coated wells in PM at 30,000 cells per cm2 for skeletal and 15,000 cells per cm2 for easy muscle differentiation in PM. After 24 h, PM was changed with differentiation medium (DM; high-glucose DMEM (Gibco), L-glutamine (Gibco), P/S and a couple of% horse serum (Gibco)) for 4–7 days. For easy muscle differentiation, DM was supplemented with 5 ng ml−1 TGFβ (Sigma) and refreshed day by day as much as day 10 of differentiation.
Movement cytofluorometry
Single cells have been suspended in fluorescence-activated cell sorting blocking buffer (FBB; 0.2 mM EDTA and 1% FBS in PBS). Cells have been incubated with 1 μl of antibody for each 1 × 105 cells for 30 min at 4 °C at nighttime with the next fluorochrome-conjugated antibodies: CD146–FITC (BioRad), CD44–PE (Biolegend), CD90–APC (Biolegend) and CD56–FITC (Biolegend) (Supplementary Desk 3) and rinsed in FBB. Cells have been analyzed (BD LSRII stream cytometer; minimal of 10,000 occasions per situation) and in comparison with unstained controls utilizing FlowJo software program.
IF
Tissue samples have been mounted (4% paraformaldehyde (PFA), 4 °C in a single day), washed in PBS, dehydrated and wax-embedded. Alternatively, samples have been snap-frozen, OCT-embedded (Sakura Finetek) after which saved at −80 °C. Sections of 5–10 mm have been minimize (MicroM Hm 325 microtome or Leica cryostat). Wax-embedded sections have been antigen retrieved (citrate buffer pH 6; Sigma-Aldrich). Sections have been permeabilized (0.5% Triton X-100 in PBS for 10 min at room temperature) earlier than quenching (30 min at room temperature with 0.1 M NH4Cl), washing and software of blocking resolution (10% BSA (Sigma-Aldrich) in PBST (PBS with 0.1% Triton X-100)) for 1 h at room temperature. Samples have been incubated with major antibodies (Supplementary Desk 3) in blocking buffer in a moist chamber in a single day at 4 °C. Samples have been washed 3 times in 0.1% PBST after which incubated with secondary antibodies at 1:200 (donkey anti-rabbit 488, donkey anti-mouse 568, donkey anti-goat 633 and donkey anti-rat 647; Supplementary Desk 3) and Hoechst 33342 (Thermo, H1399; 1:1,000) for 1 h at room temperature.
Cells have been mounted on the finish of experiments in 4% PFA at room temperature for five min, washed in PBST, permeabilized for 1 h in PBS + 0.3% Triton X-100 at room temperature and blocked in PBS + 0.3% Triton X-100 + 3% FBS for 1 h at room temperature. Cells have been incubated in a single day at 4 °C with major antibodies in PBST + 1% FBS. After washing with PBST, cells have been incubated with secondary antibodies for 1 h at room temperature. Nuclei have been counterstained with Hoechst 33342 (Thermo Fisher). Stained samples have been imaged utilizing a Zeiss LSM 710 inverted confocal microscope or Hamamatsu Nanozoomer S60 digital slide scanner.
Decellularization
Porcine esophagi have been remoted from juvenile home pigs (n = 8, 10 kg, each sexes, Royal Veterinary School). Esophagi have been dissected en bloc, with vagus nerves eliminated, washed intraluminally and extraluminally with povidone iodine and double-distilled H2O, saved at −80 °C and thawed in a single day at 4 °C the day earlier than decellularization. Mucosa was eliminated and esophagi divided into two 7–8-cm-long sections after which decontaminated in a single day at room temperature36: 320 mg l−1 gentamycin sulfate (BioChemica, PanReac AppliChem), 600 mg l−1 clindamycin hydrochloride (Sigma-Aldrich), 500 mg l−1 vancomycin hydrochloride (ApexBio) and 100 mg l−1 amphotericin B from Streptomyces sp. (Sigma-Aldrich). Esophageal sections have been decellularized utilizing a modified detergent–enzymatic remedy (DET)30,35,51. Briefly, esophagi have been cannulated with glass connectors at every finish, secured in a glass chamber and perfused utilizing a peristaltic pump (i150 C1 R3, i-pumps), over a 10-day interval. For the primary 2 days, esophagi have been washed (Milli-Q water, 9 ml min−1) renewed each 12–18 h. Esophagi underwent three consecutive DET cycles, every lasting 48 h. For every cycle, 4% sodium deoxycholate (Sigma-Aldrich) resolution was perfused for 4 h at room temperature, adopted by in a single day rinse with Milli-Q water. Subsequently, esophagi have been perfused with 25 MU of DNAse I (EMD Millipore) in 1× Hanks’ Balanced Salt Resolution (Thermo Fisher Scientific; 3 h, 3 ml min−1, 37 °C), adopted by in a single day rinse with Milli-Q water. This sequence was repeated 3 times (days 2–8; Prolonged Information Fig. 1e). After completion of three DET cycles, esophagi underwent a closing rinse with Milli-Q water for two days (9 ml min−1), with resolution renewal each 12–18 h. Decellularized esophagi have been saved in PBS supplemented with 1× antibiotic and antimycotic resolution (Gibco) and sterilized by gamma irradiation with 1.25 kGy over 12 h for 3 cycles at room temperature. This course of resulted in 16 decellularized scaffolds. Decellularized sterile organs have been saved at 4 °C earlier than recellularization (as much as 5 months). Sterility was confirmed earlier than seeding after a 72-h preconditioning interval (in a 10-cm petri dish in PM at 37 °C, 5% CO2) by microscopy, tradition and sensitivity testing.
Recellularization process
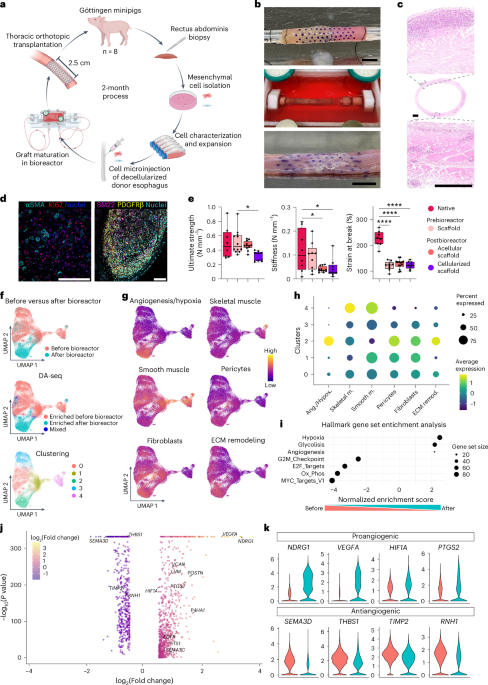
After preconditioning, decellularized scaffolds have been mounted on a stripette connected to a stepper motor, rotating by 36° to facilitate ten circumferential injections. As soon as cut up, cells have been pooled at a ratio of seven:3 MABs:FBs, with 5 × 105 reserved for stream cytometry and snRNAseq and resuspended (1 × 105 per µl) 1:1 in 15% Kind A porcine pores and skin gelatin (Sigma-Aldrich, G1890-500G) at 37 °C. Cells have been drawn up into 500-µl 29G syringes (BD) mounted in a handbook microsyringe pump (Neurostar) and injected subadventitially (30 µl per injection) at a forty five° angle to the scaffold at marked 3-mm longitudinal and transverse intervals underneath a laminar stream hood. Final graft size was roughly 3.6 cm (that’s, 360 × 106 cells per graft). As animal E solely had sufficient cells for one 3-cm scaffold, a complete of n = 15 grafts have been produced. Recellularized grafts have been positioned in custom-made bioreactors and sutured (3/0 silk) to connectors to facilitate graft rotation and intraluminal stream. Bioreactor chambers have been stuffed with 150 ml of PM + 1:100 antibiotic and antimycotic resolution (Sigma-Aldrich) and sealed. Bioreactors have been linked to a medium reservoir by Masterflex tubing and incubated (37 °C, 5% CO2). Grafts have been maintained underneath static situations in a single day, adopted by 6 days of dynamic tradition utilizing a peristaltic pump (Masterflex; 6.25 ml min−1), with simultaneous intraluminal and extraluminal perfusion. Each reservoir and bioreactor lids have been linked with 0.22-µm filters to permit gasoline alternate. Medium alternate occurred at 48-h intervals underneath sterile situations, with affirmation of sterility at intervals by microscopy and tradition. Grafts have been transported to the animal facility in T75 flasks with PM on ice. Sterility was examined immediately earlier than implantation.
Surgical strategies
Anesthetic protocol
Animals have been made nil by mouth 6 h earlier than process, with water till 1 h earlier than. Tiletamine–zolazepam (Virbac; 2 mg kg−1) and medetomidine (Chanelle; 0.02 mg kg−1) have been administered intramuscularly (IM) as a premedication. Inhalational anesthesia (3–4% isoflurane) was delivered by facemask for all procedures and the larynx was sprayed with xylocaine earlier than endotracheal intubation. Intravenous (IV) cannulation was obtained in an ear vein for thoracotomy process solely. Anesthesia was maintained with isoflurane (1.5–2.5%) with buprenorphine (Animal Care, 0.03 mg kg−1) and meloxicam (Boehringer Ingelheim, 0.4 mg kg−1) administered IV or IM. After surgical procedure, atipamezole was administered (Chanelle, 0.02 mg kg−1) and animals have been extubated on the first signal of the swallowing reflex.
Biopsy assortment
The shaved surgical web site was cleaned with chlorhexidine and povidone iodine and a 3–4-cm belly wall midline linear incision was made. A 5 × 5-mm2 part of rectus abdominis muscle and aponeurosis (fascia) was eliminated and the defect was sutured (3/0 vicryl; Ethicon, W9730). Wound closure was carried out with subcutaneous 3/0 vicryl and subcuticular 4/0 undyed vicryl (Ethicon, W9922) earlier than software of pores and skin glue (Animus).
Graft transplantation
Animals have been anaesthetized as above, with Augmentin (1000/200, Sandoz) at 30 mg kg−1 administered IV 30 min earlier than pores and skin incision. A steady IV infusion of Hartmann’s resolution (Baxter) was delivered all through. After positioning within the left lateral decubitus, the shaved surgical web site was cleaned with chlorhexidine and povidone iodine and a 10-cm, right-sided, muscle-cutting, transpleural thoracotomy was carried out by the fifth–sixth intercostal area. After isolation of the esophagus and identification of vagus nerves, a 2-cm midesophageal resection was carried out with 5-mm samples taken for mechanical testing and IF. Grafts have been transported in PM at 4 °C and washed with sterile NaCl. The proximal and distal posterior partitions of graft and native tissue have been anastomosed (5/0 Prolene, Ethicon, W8710) earlier than retrograde positioning of bioabsorbable polydioxanone (PDS) stents (10 × 8 × 10 mm3, 4 cm; SX-Ella, CZ) throughout each anastomoses, with growth in situ and securing with 5/0 PDS (Ethicon) earlier than anterior wall anastomoses. The graft was wrapped with a pleural pedicle, secured with 5/0 Prolene. The intercostal area was closed with 3/0 vicryl rib-wrapping sutures; no chest drain was used. An intercostal block was carried out with 0.5% bupivicaine (5 mg ml−1, 2 mg kg−1) earlier than closure with subcutaneous 3/0, subcuticular 4/0 vicryl and pores and skin glue.
Endoscopy
Endoscopy was undertaken utilizing a versatile GIF-XP260N ultraslim gastroscope (Olympus). Mucosal and polyp biopsies have been undertaken with 2-mm alligator-jaw biopsy forceps (Olympus, FB-220K.A) down the working channel, with specimens mounted in PFA 4%. Strictures have been dilated with an endoscopic balloon dilatator (Maxi LD 0.035-inch PTA dilation catheters, Cordis; vary: 10–16 mm) handed both parallel to the scope underneath direct laryngoscopic imaginative and prescient or over a 0.035-inch Amplatz stiff information wire (Cook dinner Medical) till it straddled the stricture. Balloon inflation was carried out underneath imaginative and prescient with manometric management utilizing an in-line strain gauge (Alliance II Built-in Inflation System, Boston Scientific) for two min earlier than repeat endoscopic inspection. The place required, stents have been repositioned endoscopically utilizing the 18 Fr, solid-olive-tip 45-cm supply system.
Postoperative care
Postthoracotomy analgesia included oral paracetamol (30 mg kg−1) as soon as day by day for five PODs, meloxicam (0.4 mg kg−1, injection or oral) on PODs 2 and 4, buprenorphine (0.01 mg kg−1) IV 6 h postoperatively and as wanted and methadone to animals D, E and G on PODs 1, 3 and three respectively. All animals obtained surgical antibiotic prophylaxis (Synulox 100 mg, BD) for five days. Animal D was given a further IM dose of depocillin on POD 2 due to gradual restoration and animal F was given prolonged prophylaxis (IM Baytril, 5 mg kg−1) on PODs 5–9 due to a temperature of 39.2 °C after esophageal international physique removing on day 3. Omeprazole (20 mg as soon as day by day) was administered in feed as per ERNICA tips after EA restore. Oral prednisolone was administered to animals as soon as day by day in batch 1 from POD 39 (animals A and C) and POD 46 (animal B), after histology from polyps indicated hyperepithelialization with edema, till 13 days after stent alternate. Subsequently, prednisolone was administered prophylactically in animals E and G on POD 12 and animals F and H on POD 19 till 30 days after deliberate stent alternate.
Postoperatively, animals had entry to water ad libitum for twenty-four h, adopted by liquid hypercaloric feed (moist, crushed SMP pellets supplemented with Complan (Nutricia)) for 1 week and semiliquid meals (moist mash basal food plan) for week 2 earlier than recommencing regular food plan.
Deliberate humane endpoints
As per our license, culling was mandated if the animal demonstrated a change in regular habits and/or physiology and it was felt unlikely to make passable enchancment inside 72 h following an intervention. Adversarial results together with stent migration and stricture have been anticipated, with preliminary authorization to deal with these a most of 3 times endoscopically for instance, balloon dilation (allowed twice) and stent adjustment or substitute (allowed as soon as). Nonetheless, early FB obstruction required endoscopy in two of the primary 4 animals, leading to extra endoscopies at early time factors than anticipated. Subsequently, license amendments have been accredited to permit for as much as seven unplanned diagnostics with or with out therapeutic endoscopies, together with the necessity for balloon dilation, biopsy or debridement of polyps, international physique removing, stent repositioning or reapplication, mandating culling within the presence of a ‘symptomatic stricture recurrence not amenable to endoscopic intervention’.
Animal culling
Premedication and normal anesthetic induction have been as described above. Endoscopy and HRIM have been undertaken (see beneath) to evaluate esophageal peristalsis. On completion, sodium pentobarbitone (140 mg kg−1) was administered IV adopted by exsanguination. Esophagi have been explanted en bloc and transported in HypoThermosol FRS (StemCell Applied sciences) for processing.
HRIM
Anesthetized animals on the 3-month and 6-month endpoints (n = 7) underwent HIRM (Photo voltaic GI HIRM system, Medical Measurement Methods) and a 6-F solid-state catheter (Unisensor) with 26 1-cm-spaced high-resolution strain sensors and 12 impedance channels. The catheter was inserted by the oropharynx underneath direct laryngoscopy and superior underneath mixed endoscopic and fluoroscopic steering, till a minimum of three strain sensors reached the abdomen. The manometric channels similar to the graft have been recognized radiologically following endoscopic visualization of graft sutures and the gastroesophageal junction. After baseline readings, the esophagus was insufflated with air and water boluses (20, 40 and 60 ml) to each proximal and distal esophagus by a further oropharyngogastric catheter, with the variety of aliquots depending on secondary peristalsis response (minimal of two per situation). Information have been analyzed utilizing Manometry and Evaluation Software program model 10.0 (Medical Measurement Methods).
Organ tub contractility
Tissue samples (circumferential rings from native proximal, graft and two sequential distal areas) have been transferred to oxygenated Krebs resolution at room temperature. Rings (2 mm large) have been opened and particular person strips (~10 × 2 mm2) remoted and mounted in organ baths (10 ml; SI-MB4, World Precision Devices). Samples have been linked to power transducers (SI-KG20, World Precision Devices) by 4.0 sutures (High quality Science Instruments) underneath an preliminary stress of 1 g. Organ baths have been maintained at 37 °C and obtained periodic perfusion of oxygenated Krebs resolution. After 1 h of equilibration, contractile exercise was recorded utilizing a Lab-Trax-4 information acquisition system (World Precision Devices). Tissue samples have been subjected to trains (40 V, 0.3-ms pulse period) {of electrical} subject stimulation (EFS) for 10 s, each 1 min, by platinum electrode loops situated at each ends of the tissue pattern utilizing a MultiStim System (D330, World Precision Devices). Graded responses to EFS have been assessed at 5, 10 and 20 Hz (5 stimulations at every). After closing software of EFS, tissues have been allowed to recuperate for 10 min earlier than CCh software (1 mM and 10 mM) separated by washout and 10-min restoration. Samples have been then mounted and sectioned. Contractility information have been analyzed utilizing Labscribe model 4 software program (World Precision Devices), together with response to EFS (space underneath the curve (AUC) at some point of EFS) and maximal contractile amplitude in response to CCh. To account for tissue variability, uncooked responses have been normalized to moist tissue weight (g). Consultant traces have been compiled in Plot2.
Histology
First, 0.5-cm-long tissue rings have been sampled from native and graft esophagus instantly after harvest, from decellularized and gamma-irradiated esophagus and from recellularized scaffolds earlier than transplantation. Samples have been mounted in 4% PFA at 4 °C, dehydrated in a single day, paraffin-wax-embedded and minimize into 5-µm sections or snap-frozen in liquid nitrogen, embedded in OCT and minimize into 7-µm sections. For chemical staining, slides have been stained with hematoxylin and eosin (H&E; Thermo Fisher/Leica), Masson’s trichrome (MT; one-step MT) or elastic van Gieson (EVG; Atom Scientific) in response to customary working procedures of the Histopathology Division, Nice Ormond Avenue Hospital (GOSH).
Immunohistochemistry
Antibodies have been optimized on 3-µm pig esophageal tissue sections. Immunohistochemistry (IHC) was carried out on a Leica Bond RX automated platform (Leica Biosystems). In short, antigen retrieval was undertaken to unmask epitopes (heat-induced epitope retrieval (HIER), Bond protocol F). Endogenous exercise was blocked with peroxidase utilizing a Bond polymer refine equipment (Leica Biosystems, DS9800). Slides have been incubated with major antibodies to nNOS (Sigma, AB5380; 1:1,000 dilution, HIER1 for 20 min), STMN2 (Thermo Fisher, 67204-1-Ig; 1:500 dilution, HIER2 for 30 min) and PGP9.5 (BioRad, 7863–2004; 1:10,000 dilution, HIER1 for 20 min). Subsequent, a postprimary antibody was utilized with horseradish peroxidase (HRP)-labeled polymer, adopted by DAB chromogen resolution (all a part of the Bond polymer refine equipment). Sections have been counterstained with hematoxylin, washed, dehydrated in graded alcohols, cleared in two xylene adjustments and mounted. Imaging was undertaken utilizing a 3DHistech P480 with ×40 goal. Subsequent evaluation was carried out with QuPath (model 0.2.3; and/or Leica ImageScope (Leica Biosystems) software program.
Biochemical quantitative evaluation
Tissue was sampled from native instantly after harvest and from decellularized or gamma-irradiated esophagus.
Quantification of DNA
DNA was extracted from tissue (~25 mg) utilizing a DNeasy blood and tissue equipment (Qiagen) and quantified by a NanoDrop 1000 spectrophotometer (Thermo Fisher Scientific). Complete DNA was normalized to the tissue mass individually in three technical replicates.
ECM quantification
ECM proteins have been extracted from moist tissue (5–20 mg). Complete collagen was extracted with a Sircol soluble collagen assay equipment (Biocolor). Tissue was finely diced, dried and weighed. Complete elastin was extracted with the Fastin elastin assay (Biocolor). Complete sulfated glycosaminoglycan (GAG) was extracted with Blyscan sulfated GAG assay equipment (Biocolor). Complete ECM protein focus was decided utilizing the usual curve, earlier than being normalized to the tissue mass and calculating the imply and s.d. from three technical replicates.
Residual galactose-α-1,3-galactose detection
IHC staining of paraffin sections of native and decellularized tissues was carried out utilizing a major antibody of galactose-α-1,3-galactose (αGal) epitope (M86) monoclonal antibody (ALX-801-090-1, Enzo Life Sciences) (1:5) and DAKO (Animal Analysis Equipment, K3954) with Mayer’s hematoxylin as a nuclear counterstain. Optical densities of the obtained gentle microscopy IHC photographs have been evaluated utilizing Picture J-IHC toolbox. For quantitative evaluation, native (n = 4) and decellularized (n = 6) samples have been weighed, washed with PBS, homogenized and centrifuged; supernatants have been collected for αGal ELISA. Briefly, 100 µl of supernatant was incubated with αGal epitope (M86) monoclonal antibody (Enzo Life Sciences) (1:250) for two h. After washing nonbinding antibodies, secondary antibody goat anti-mouse HRP-linked immunoglobulins (p0447) (1:1,000)) have been utilized for 1 h. The secondary antibodies have been visualized utilizing TMB substrate for 10 min and the response was stopped utilizing 1 N HCl. Absorbance was measured at 450 nm utilizing a microplate reader. αGal epitopes (Ludger, CAA-ALPHAGAL-01) and PBS have been used as constructive and unfavorable controls, respectively.
Biomechanical checks
Tensile checks evaluated esophagi and grafts circumferentially. Ring-shaped specimens have been used for pretransplantation testing (Fig. 1e), whereas rectangular specimens have been used after transplantation due to restricted tissue availability (Fig. 3c). Samples have been stretched at a uniform fee till break level. For rectangular samples, the preliminary gripper distance (D) was set at 10 mm, with tissue width (W) of ~2 mm and thickness (t) of ~4 mm. Ring samples had a size (L) of 5 mm and diameter of ~10 mm. A preload of 0.01 N was utilized to all samples, with a stretching pace 10 mm min−1. Stiffness was decided as slope of the linear match of the power–displacement curve over the preliminary 5-mm elongation. Outliers in each in vitro and in vivo testing have been recognized by the ROUT outlier check (α = 0.05) and excluded appropriately. Final energy was calculated by dividing the utmost load (Tmax) by both the width (W) of the oblong pattern or twice the size (2L) of the ring pattern. Pressure at break was outlined as the utmost pressure within the checks. Biomechanical properties have been assessed in in vitro specimens in native samples (n = 8), postbioreactor acellular scaffolds (n = 14) and postbioreactor cellularized samples (n = 9). Due to tissue availability limitations, prebioreactor scaffolds have been analyzed in n = 13 samples for final energy however n = 9 samples for stiffness and pressure at break. Ex vivo, biomechanical properties have been assessed in seven of eight animals due to the timing of dying of animal D.
Transcriptomic evaluation
Pattern processing
Samples have been saved in HypoThermosol FRS at 4 °C. Inside 24 h, tissue samples have been faraway from HypoThermosol FRS and extra liquid was eliminated. Samples have been snap-frozen in isopentane and dry ice earlier than being embedded in OCT on dry ice. OCT samples have been cryosectioned on a Vivid OTF5000 cryostat. In whole, 15 25-µm sections of recellularized esophageal scaffold have been saved at −80 °C for isolation of nuclei. Samples included ‘native’ esophagus (n = 4), eliminated at (1) organ harvest for decellularization in donor home pigs (n = 3) or (2) in minipigs at time of transplantation (n = 1, animal C) as constructive controls, a recellularized scaffold after 1 week of bioreactor maturation (‘preimplantation graft’) as a unfavorable management (n = 1, animal C) and grafts at 1 month (n = 1, animal D), 3 months (n = 2, animals E and F) and 6 months (n = 3, animals A, B and C) after transplantation.
ST library preparation
First, 10-µm sections have been loaded onto Visium Spatial Gene Expression slides as per protocol CG000240 (1000184, 10X Genomics). In parallel, complete cross-sections of the esophagi have been beforehand methanol-fixed, stained with H&E and imaged as per protocol GC000160 on a Motic EasyScan One slide scanner at ×40 magnification to pick out probably the most consultant portion for the Visium slide. Loaded Visium slides have been subsequently mounted, H&E-stained and imaged. Sections have been permeabilized (12 min) for mRNA seize and reverse-transcribed earlier than present process second-strand synthesis to generate full-length complementary DNA, adopted by library preparation (protocol CG000239). The permeabilization time was established as acceptable utilizing the Visium spatial tissue optimization equipment (1000193, 10X Genomics) following 10X Genomics protocols (CG000240 for sectioning and slide loading, GC000160 for H&E staining and brightfield imaging and CG000238 for mRNA seize, reverse transcription, second-strand synthesis and fluorescence imaging).
Single-nucleus transcriptomic library preparation
Cryopreserved aliquots of the autologous cells used to recellularize scaffold (animal C) have been revived and checked for focus (500,000 cells) and viability (94.6%) utilizing an acridine orange–propidium iodide (AO/PI) stain on a Luna FL automated cell counter. Cells have been pelleted by centrifugation at 300g for five min at room temperature. The supernatant was eliminated and the dry pellet was snap-frozen.
Nuclei have been remoted from 25-µm sections of recellularized scaffold talked about beforehand and snap-frozen pelleted cell combine, utilizing the Chromium nucleus isolation equipment (1000494, 10X Genomics; protocol CG000505), treating the cell pellet in the identical method because the tissue.
Remoted nuclei have been checked for focus utilizing an AO/PI stain on a Luna FL automated cell counter. A complete of 20,000 nuclei for every pattern have been used to generate single-nucleus transcriptomic libraries with a 10X Genomics Chromium controller utilizing the Chromium next-GEM Chip Okay and Chromium next-GEM single-cell 5′ v2 kits (1000287 and 1000265, respectively, 10X Genomics; protocol CG000331).
Sequencing
Ensuing single-nucleus and ST libraries have been sequenced on an Illumina NextSeq 2000 sequencer, utilizing a NextSeq 2000 P3 (100 cycles) sequencing equipment.
Single-nucleus and ST computational evaluation strategies
Filtered gene expression matrices produced by Cell Ranger and House Ranger (aligned towards the ENSEMBL Sus scrofa reference genome construct model 11.1) have been filtered for empty droplets by cellbender. Multiplexed libraries have been demultiplexed utilizing vireoSNP and filtered for possible doublets. Fastq information, gene expression matrices, House Ranger outputs, and BAM and VCF information (for demultiplexing) can be found from the Gene Expression Omnibus (GEO) underneath accession quantity GSE280737. Subsequent evaluation was carried out utilizing Seurat (model 4.4.0) inside Docker (model 26.1.4, construct 5650f9b), with the pipeline out there on-line (https://doi.org/10.5522/04/27303705).
All single-nucleus samples have been filtered utilizing handbook thresholds for doublets (higher bounds on the variety of molecules and genes per droplet) after which normalized utilizing NormalizeData, FindVariableFeatures and ScaleData. A principal part evaluation discount was calculated and corrected for interlane batch results utilizing Concord. Sequencing expertise and lane quantity have been used as latent variables within the logistic regression framework inside Discover(All)Markers. Custom_FindMarkers used the identical methodology when evaluating steady metadata. GSEA was carried out with fgsea utilizing the Hallmark gene units for S. scrofa. Differential abundance evaluation was carried out utilizing DA-seq. Clustering decision was decided by Clustree32 as one of many largest resolutions earlier than clusters started merging. Clustree was additionally used to find out resolutions for subclustering. Expression of units of genes was calculated utilizing Seurat’s AddModuleScore. Warmth maps and dot plots have been generated utilizing gene expression, scaled throughout samples. For ST samples, spots disconnected from the principle tissue slice or the place tissue was folded have been eliminated computationally. Expression was normalized with SCTransform. The dominant cell kind for every spot was decided by correcting for intersample variations with Concord, then clustering outcomes and assigning a cell kind identification to every cluster utilizing anatomical location and the expression of marker genes. In single-nucleus volcano plots, P values have been constrained to a minimum of 1 × 10−330. Spatial similarity between samples was quantified by counting the variety of neighboring spots for every pair of cell sorts after which utilizing them to compute Euclidean distances between samples (visualized with circlize).
Statistics and reproducibility
Pattern dimension n refers back to the variety of unbiased experiments or organic replicates, as acknowledged within the determine legends. Within the in vitro portion of the research, the 2 decellularized esophagi from the identical animal have been handled as particular person experimental replicates as they underwent unbiased decellularization and recellularization (n = 16 and 15, respectively). In vivo, ‘animal’ denotes one organic replicate. If information displayed a possible outlier, this was examined utilizing ROUT’s outlier check and excluded as acceptable (Supply Information). Except in any other case acknowledged, information are displayed as box-and-whisker plots, displaying the median, interquartile vary (IQR) and minima and maxima (vary), with all replicates proven as particular person factors. Animal survival was plotted as a Kaplan–Meier survival plot. Statistical significance was decided utilizing paired or unpaired t-tests for comparability of parametric information between two totally different experimental teams or an bizarre one-way evaluation of variance (ANOVA) with Tukey’s multiple-comparison check for evaluation in additional than two teams. A P worth of <0.05 was thought-about statistically important (*P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001). Statistical evaluation was carried out utilizing GraphPad Prism model 10.4.0.
Reporting abstract
Additional data on analysis design is out there within the Nature Portfolio Reporting Abstract linked to this text.









Leave a Reply